11 minutes ago 0
A white tiger already feels unforgettable, but in original scratch art it becomes something even more powerful:...
Your Guide to Money & Cash Flow
 A White Tiger – If You Have Never Seen One, Here’s Your Chance! An Original Scratch Art On A 14″X18″X1/8″ Board – One Of A Kind! – Www.luzimmscratchart.com
A White Tiger – If You Have Never Seen One, Here’s Your Chance! An Original Scratch Art On A 14″X18″X1/8″ Board – One Of A Kind! – Www.luzimmscratchart.com  Chakra Bracelet: Meaning, Benefits & How to Wear One
Chakra Bracelet: Meaning, Benefits & How to Wear One  Traditional Scottish Cranachan Recipe
Traditional Scottish Cranachan Recipe 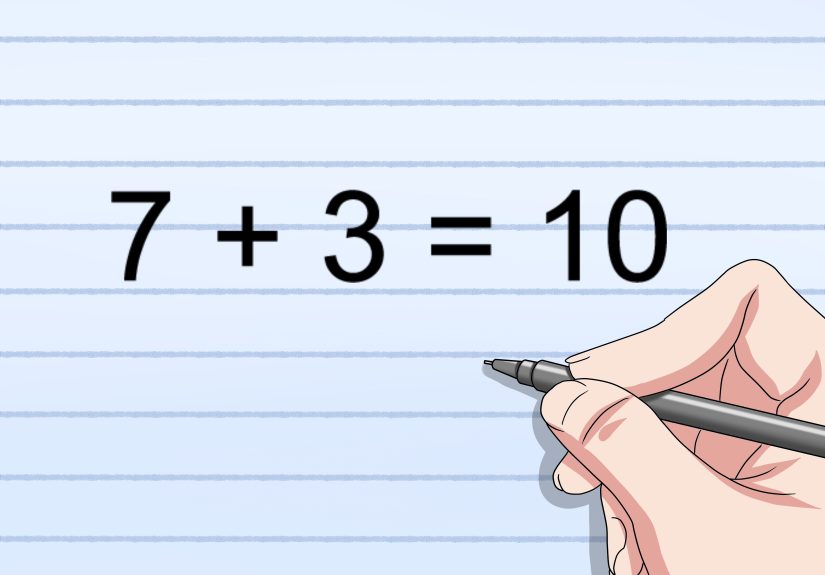 4 Ways to Determine the Reading Level of a Book
4 Ways to Determine the Reading Level of a Book  The Switch 2 Pokemon Legends: Z-A Bundle Is Back in Stock for Black Friday
The Switch 2 Pokemon Legends: Z-A Bundle Is Back in Stock for Black Friday