Table of Contents >> Show >> Hide
- What ECMO Is (and What It Isn’t)
- How the ECMO Circuit Works
- Types of ECMO: VV vs VA (Why Two Letters Matter a Lot)
- When Is ECMO Used?
- A Concrete Example: What ECMO “Looks Like” in Decision-Making
- Day-to-Day Life on ECMO in the ICU
- Risks and Complications: Why ECMO Is “High-Reward, High-Responsibility”
- How Long Does ECMO Last?
- What “Success” Means on ECMO
- Questions Families Can Ask (That Actually Help)
- After ECMO: Recovery Is Often the Second Big Chapter
- on Real-World Experiences with ECMO
- Conclusion
ECMO is the ICU equivalent of calling in a pit crew for your heart and lungs: not a cure, not a magic reset button,
but a way to keep oxygen moving and carbon dioxide leaving while your body gets a fighting chance to recover.
If you’ve heard it described as a “heart-lung machine,” that’s not wrongjust incomplete. ECMO is less like a replacement
organ and more like a very intense, very temporary detour around organs that are too injured or exhausted to do their job.
Because ECMO is used when someone is critically ill, it can sound terrifying (it is serious) and futuristic (it is also
surprisingly mechanical). Let’s translate the jargon into real life: what ECMO does, when it’s used, what the different
types mean, what the risks are, and what patients and families can realistically expectday by day, alarm by alarm.
What ECMO Is (and What It Isn’t)
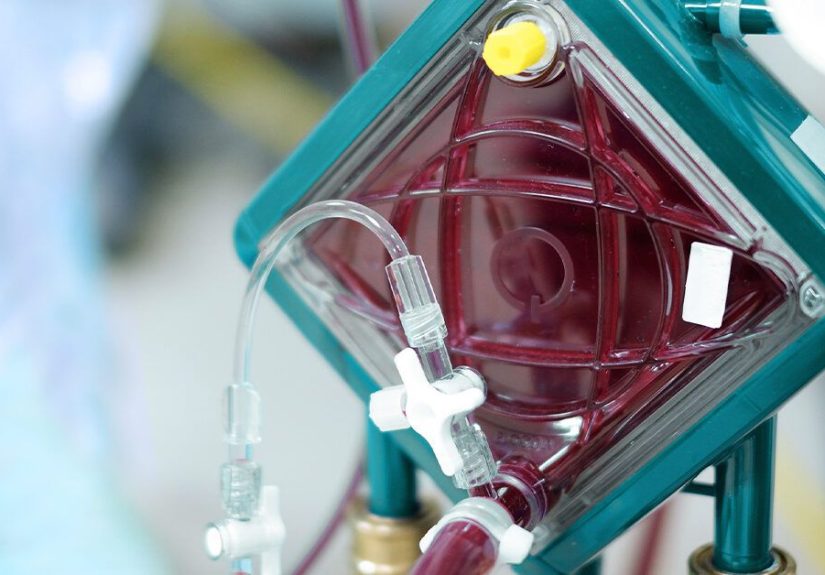
Extracorporeal membrane oxygenation means oxygenating blood outside the body.
In practice, ECMO is a circuit of tubing connected to large blood vessels. Blood leaves the body through a cannula (a
large tube), passes through an artificial “lung” (the membrane oxygenator) where oxygen is added and carbon dioxide is removed,
and then returns to the body warmed to body temperature.
Here’s the key mindset shift: ECMO buys time. It supports oxygen delivery and/or circulation so that
the underlying problemsevere pneumonia, ARDS, heart failure, cardiogenic shock, post-surgery heart dysfunction, and morecan
be treated while the heart and lungs rest. ECMO can also be used as a “bridge” to a decision, recovery, or transplant.
What ECMO is not: it’s not the same as a ventilator, and it’s not the same as long-term artificial heart support.
Ventilators move air in and out of the lungs; ECMO oxygenates blood directly. ECMO is typically intended for short-term support
(often days to weeks, sometimes longer in select scenarios), not permanent therapy.
How the ECMO Circuit Works
1) Cannulas: the “on-ramp” and “off-ramp”
Cannulas are placed into large veins and/or arteriescommonly in the neck (internal jugular), groin (femoral vessels),
or sometimes the chest depending on the situation. Placement usually happens under anesthesia or deep sedation and is guided
by imaging and careful monitoring.
2) The pump and the oxygenator: an external heart + external lung
The pump moves blood through the circuit. The membrane oxygenator does gas exchangeadds oxygen and removes carbon dioxide.
Many systems also include a heat exchanger to keep blood at an appropriate temperature before it returns to the body.
3) Anticoagulation and constant monitoring
Blood is designed to clot when it touches “foreign” surfaces. ECMO circuits are, by definition, foreign surfaces. So teams
often use blood thinners (anticoagulation) and frequent labs to reduce the risk of clots in the circuitwhile also trying
not to tip the patient into dangerous bleeding. This balancing act is one of the reasons ECMO requires a highly specialized
ICU team and round-the-clock attention.
Types of ECMO: VV vs VA (Why Two Letters Matter a Lot)
VV ECMO (veno-venous): lung support
Venovenous (VV) ECMO drains blood from a vein and returns it to a vein. It primarily supports the lungs by
oxygenating blood and removing carbon dioxide. The heart still does the main work of pumping blood around the body, but the
lungs get a chance to rest and heal with gentler “lung-protective” ventilator settings.
VV ECMO is most often used in severe respiratory failure, such as advanced ARDS, severe pneumonia, influenza, and other causes
of life-threatening low oxygen levels that don’t improve despite optimal ventilation and medical therapy.
VA ECMO (veno-arterial): heart + lung support
Venoarterial (VA) ECMO drains blood from a vein and returns it to an artery. This supports both oxygenation
and circulationuseful when the heart cannot pump enough blood to meet the body’s needs (for example, cardiogenic shock).
Because VA ECMO involves the arterial system, it can carry different risks (including limb circulation issues depending on
cannulation strategy) and requires especially careful hemodynamic management.
What about hybrid setups?
Real ICU life is messy. Some patients have mixed heart-and-lung failure. Specialized centers may use hybrid approaches
(variations in cannulation configuration) tailored to a patient’s physiology. For most readers, the takeaway is simple:
VV = lungs; VA = heart + lungs.
When Is ECMO Used?
ECMO is generally considered when conventional treatments aren’t enoughthink maximal ventilator support, medications to support
blood pressure and heart function, and other advanced ICU therapies. It is reserved for severe, life-threatening failure because
it is invasive, resource-intensive, and not risk-free.
Severe respiratory failure (often ARDS)
- ARDS from pneumonia, aspiration, severe inflammation, or trauma
- Severe viral infections (e.g., influenza, COVID-era practice in appropriate candidates)
- Life-threatening asthma or other causes of extreme carbon dioxide retention in select cases
Cardiac failure and cardiogenic shock
- Cardiogenic shock after a major heart attack
- Severe myocarditis (inflammation of the heart muscle)
- Post-cardiac surgery failure (sometimes called post-cardiotomy shock)
- Bridge support while evaluating longer-term mechanical support or transplant candidacy
Bridge-to-transplant or bridge-to-decision
In specialized programs, ECMO may support oxygenation and circulation while a patient awaits lung or heart transplantor while
a team determines whether recovery is likely, whether transplant is feasible, or whether goals of care should change.
Pediatric and neonatal use (a different world, same concept)
In newborns and children, ECMO can be used for severe respiratory or cardiac failure when other measures fail. Neonatal examples
include severe meconium aspiration, pulmonary hypertension, and congenital diaphragmatic herniaconditions where lungs and circulation
can be profoundly stressed early in life.
A Concrete Example: What ECMO “Looks Like” in Decision-Making
Imagine a patient with severe pneumonia who develops ARDS. Despite careful ventilation strategies, paralysis, prone positioning,
and optimized medications, oxygen levels remain dangerously low. At that point, the ICU team may consult an ECMO service.
If the patient is an appropriate candidate, VV ECMO can provide oxygenation while the lungs rest, allowing time for antibiotics,
anti-inflammatory strategies, and supportive care to work.
In a different scenariosay, a patient with a massive heart attack and shockVA ECMO might support circulation while cardiologists
treat blocked arteries and the heart recovers (or while the team evaluates next steps like additional mechanical support).
Day-to-Day Life on ECMO in the ICU
“Lung rest” and ventilator strategy
Many ECMO patients remain on a ventilator, but the goal is often to reduce ventilator intensity to limit further lung injury.
ECMO can offload gas exchange so the care team can use gentler settings and focus on recovery.
Sedation, comfort, and sometimes “awake ECMO”
Some patients are deeply sedated early onespecially during cannulation and unstable periods. Others may be lightened as they stabilize.
In select cases at experienced centers, patients can be awake, communicate, and even participate in physical therapy while on ECMO,
particularly in bridge-to-transplant situations. It’s not the norm everywhere, and it depends heavily on safety and stability.
Monitoring is not a vibeit’s the job
Expect frequent blood tests, imaging (like X-rays), continuous vital sign monitoring, and constant adjustments.
ECMO specialists, ICU nurses, respiratory therapists, perfusionists, and physicians coordinate care like a relay team that never stops running.
Mobility and rehab start earlier than most people think
Once stable, many teams try to prevent severe deconditioning. That can mean passive range-of-motion exercises, sitting up in bed,
and sometimes standing or walking with extensive support. Small milestones matter a lot: sitting at the edge of the bed,
taking a few steps, or even staying awake through a conversation.
Risks and Complications: Why ECMO Is “High-Reward, High-Responsibility”
ECMO can be lifesaving, but it is not gentle. Complications can come from the patient’s underlying illness, the need for anticoagulation,
the presence of large cannulas, and the fact that blood is flowing through an external circuit.
Bleeding
Bleeding is one of the most common and feared complications. Patients may bleed at cannula sites, surgical sites, or internally.
The risk exists because blood thinners are often needed to reduce circuit clottingand because critically ill patients can develop
complex clotting abnormalities even before ECMO starts.
Stroke and neurologic injury
Stroke risk can be related to bleeding (hemorrhagic stroke) or clots (ischemic stroke). Teams reduce risk through meticulous anticoagulation
management, monitoring, and rapid response to any concerning signs.
Clots in the circuit
Even with anticoagulation, clots can form in the circuit. If they compromise safety or oxygenator performance, components may need to be exchanged.
This is one reason ECMO is managed in specialized centers with trained staff and protocols.
Infection
Large cannulas, ICU length of stay, and critical illness raise infection risk. Strict sterile technique, line care, and vigilant monitoring
are central to ECMO management.
Limb circulation problems (more common concern in some VA ECMO setups)
If arterial cannulation involves the groin, blood flow to the leg can be affected. Many centers use specific strategies and monitoring
to protect limb perfusion, but it remains a known risk.
Kidney injury and multi-organ strain
Many ECMO patients are critically ill with multi-organ stress. Some require dialysis (continuous renal replacement therapy), nutritional support,
and other ICU interventions while on ECMO.
How Long Does ECMO Last?
There isn’t one timeline. ECMO duration depends on how quickly the underlying problem improves. Some patients need ECMO for a few days;
others need weeks. In specialized circumstancesespecially bridge-to-transplantsupport can extend longer, but it requires intensive planning
and constant reassessment of goals and progress.
What “Success” Means on ECMO
Success isn’t just “survived ECMO.” Clinically, teams look for signs the heart and/or lungs are taking back their workload:
improving oxygenation on lower ECMO settings, better lung compliance, stronger heart function on imaging, and stable blood pressure with fewer medications.
Weaning involves gradually reducing ECMO support while closely watching how the patient responds. If the body maintains stability, the patient may be
decannulated (cannulas removed). After that, recovery continuesoften with rehabilitation, follow-up care, and sometimes long-term support for lingering weakness
or lung/cardiac limitations.
Questions Families Can Ask (That Actually Help)
- Which type of ECMO is being used (VV or VA), and what organs are being supported?
- What is the main goal: bridge to recovery, bridge to transplant, or bridge to decision?
- What signs would indicate improvementand what signs would worry you?
- What complications are you watching most closely for in this specific case?
- How will sedation, comfort, and communication be handled?
- What does rehab look like while on ECMO and after ECMO?
- How often will the team provide updates, and who is the best point of contact?
After ECMO: Recovery Is Often the Second Big Chapter
Even when ECMO goes “well,” many patients face a long recovery. ICU-acquired weakness is common after prolonged critical illness.
Some patients need respiratory therapy, physical therapy, and gradual rebuilding of strength and stamina. Emotional recovery matters too:
ICU stays can be disorienting, and some people experience anxiety, depression, or post-traumatic stress symptoms afterward.
The good news: many patients improve substantially with time, rehab, and follow-up. The realistic news: progress can be uneventwo steps forward,
one step backand that’s still progress.
on Real-World Experiences with ECMO
If you ask clinicians what ECMO feels like, you’ll often get the same answer delivered with a tired smile: “It’s intense.”
That intensity shows up differently depending on where you’re standingat the bedside, inside the bed, or behind the machines.
Here are the most common “experience truths” people describe, woven together from typical ICU realities (not any one person’s private story).
For families: The first experience is usually sensory overload. The room is busy. Monitors beep. Pumps hum.
People come in and out with purpose, speaking in acronyms like they’re speed-running a medical dictionary. Families often describe an early moment
of shock: seeing loved ones connected to thick tubing can feel unreal, like the body has become part of a life-support ecosystem.
But then routines appeardaily rounds, regular updates, familiar faces on the nursing teamand the chaos becomes a pattern you can hold onto.
Many families anchor to small wins: “oxygen numbers improved,” “less medication,” “he opened his eyes,” “she squeezed my hand.”
Those tiny milestones can feel bigger than birthdays.
For patients: Experiences vary widely because sedation levels differ. Some patients remember little to nothing until after ECMO.
Others remember fragments: a voice, a light, a sense of pressure, the strange feeling of time moving without clear days.
Patients who are awake or lightly sedated often describe two competing realities: gratitude that they’re alive and frustration at the limits of their body.
Even communicating can be hardbecause of breathing tubes, weakness, or fatigue. In those cases, a whiteboard, hand squeezes, or simple yes/no cues
become powerful. The emotional landscape can swing fast: calm one hour, overwhelmed the next. That doesn’t mean someone is “not coping.”
It means they’re human in a situation that is objectively a lot.
For the care team: ECMO work is a constant balancing act. Staff talk about “zooming in” and “zooming out.”
Zoom in: anticoagulation levels, circuit pressures, oxygenator performance, cannula position, subtle changes in neurologic exam.
Zoom out: is the heart recovering, are the lungs improving, are we moving toward our goal? Clinicians often describe a particular kind of hope on ECMO:
it’s not blind optimismit’s data-driven hope. A slightly better chest X-ray, improved blood gases, less vasopressor support.
The team celebrates quietly, then gets back to work, because ECMO rewards vigilance.
The shared experience: ECMO tends to compress life into the present tense. People stop thinking in weeks and start thinking in shifts:
“How did today go?” Over time, many families and survivors say they remember two things most: the fear at the start, and the steady commitment of the ICU team.
And when recovery beginswhen machines are turned down, when the cannulas come out, when rehab startsit often feels like stepping out of a storm.
Not instantly “fine,” but finally moving forward.
Conclusion
ECMO sits at the intersection of advanced technology and very human urgency. It’s used when the heart and/or lungs can’t keep up and time is running out.
The goal is simple to say and hard to achieve: keep the body alive and stable long enough for recovery, transplant, or a clear decision about next steps.
If you’re facing ECMO as a patient or family member, it’s okay to feel overwhelmedask direct questions, focus on the next milestone, and remember that
ECMO care is built around a team whose entire job is to manage the scary details so you don’t have to.






